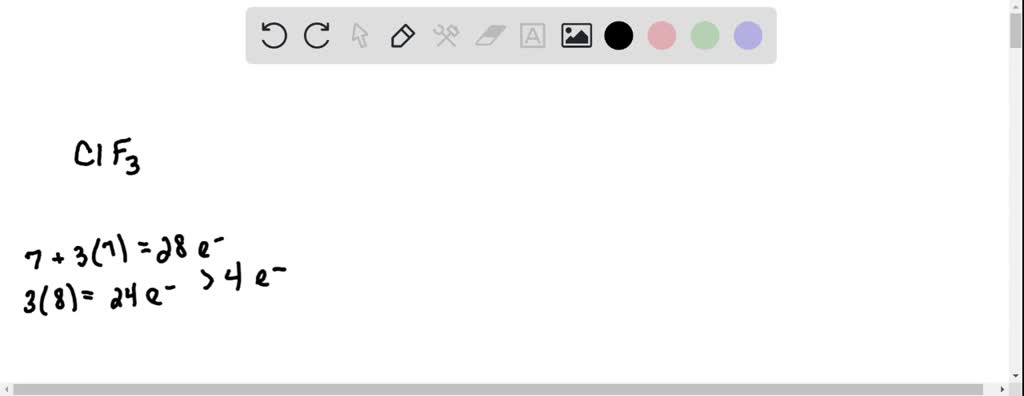
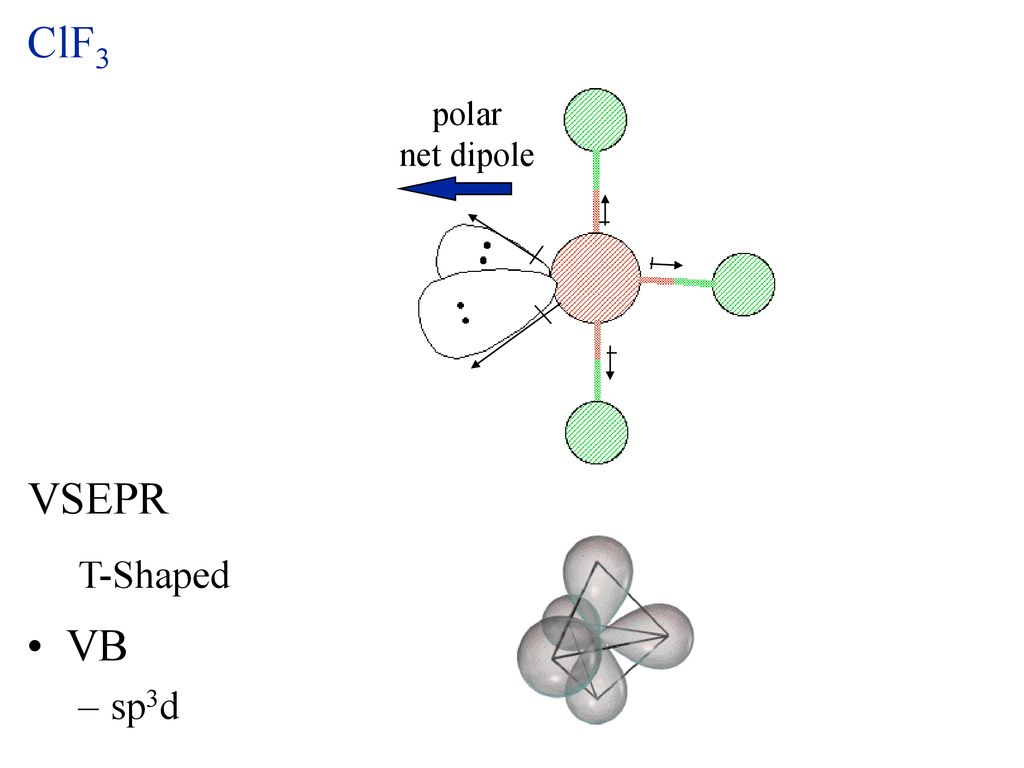
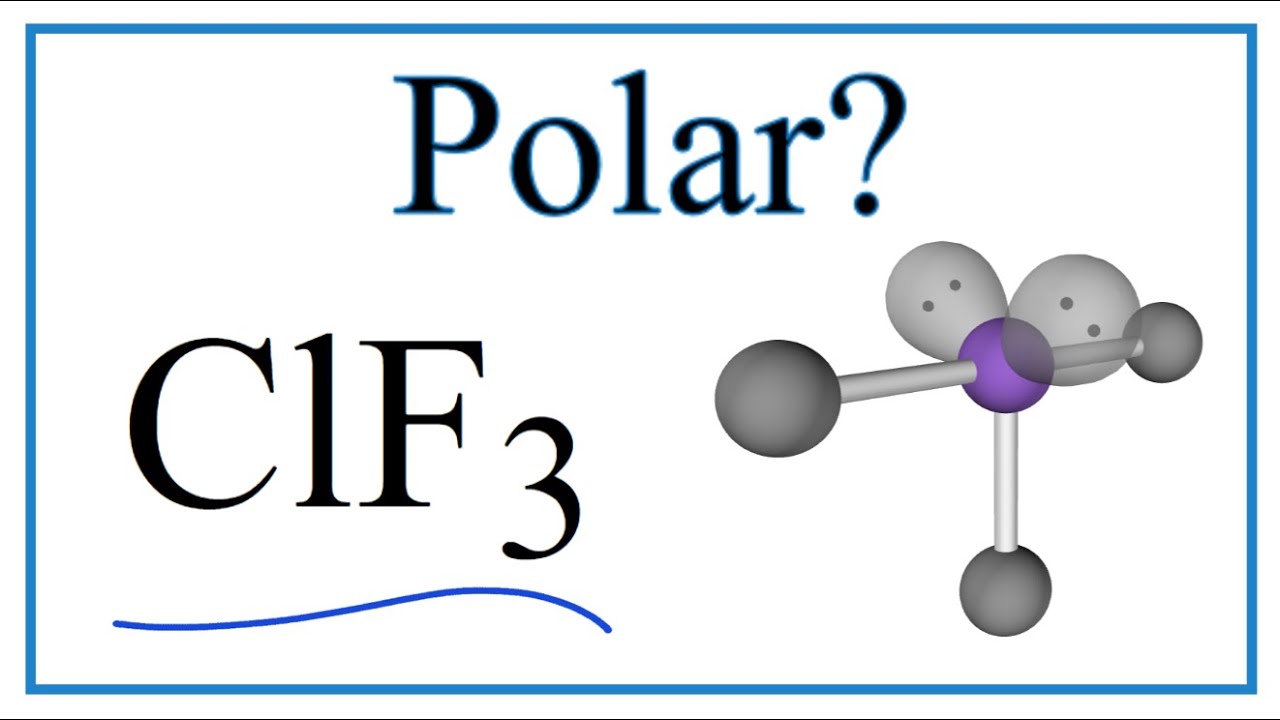
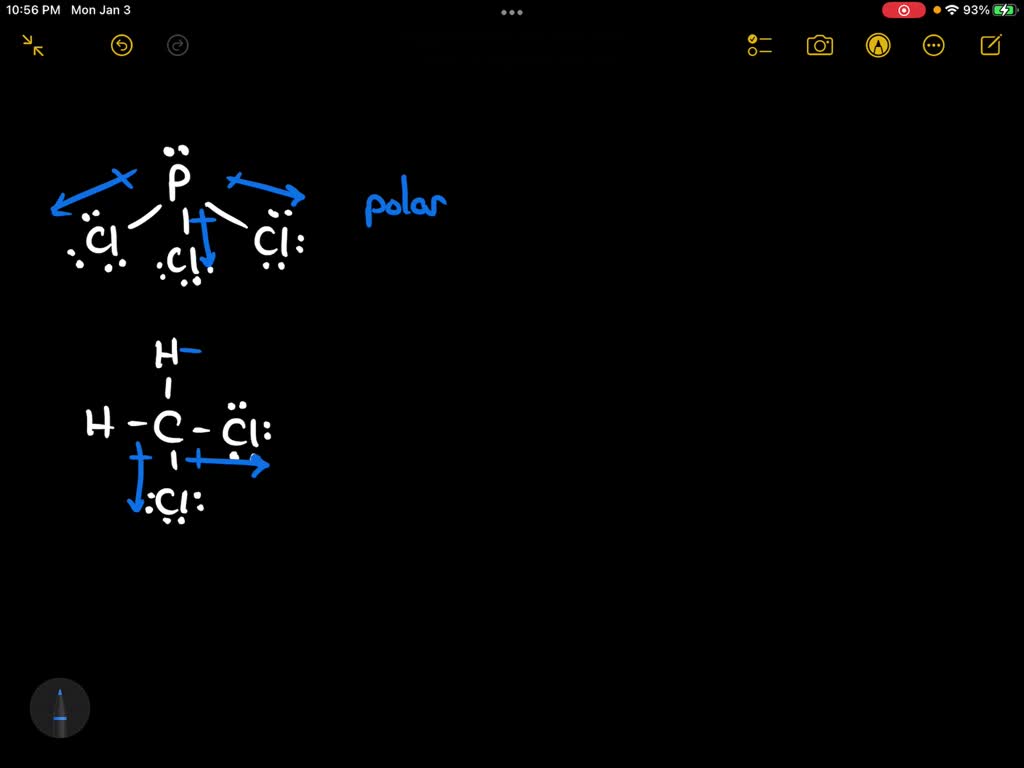
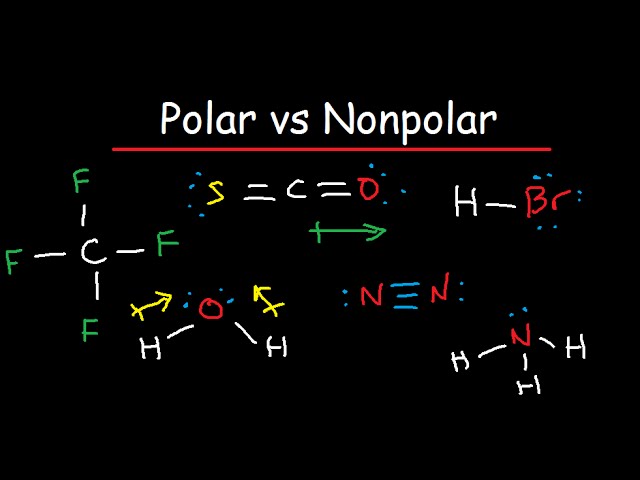
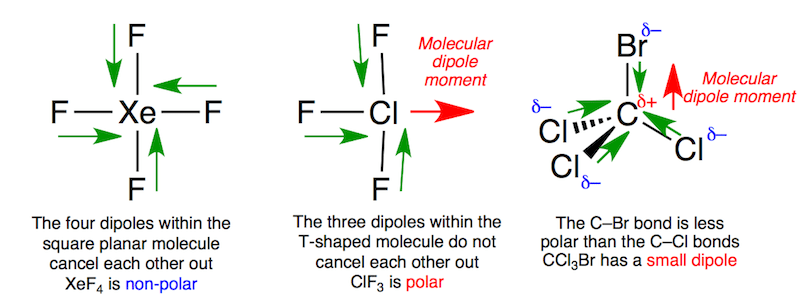
Draw the Lewis structure for ClF3. Determine its electron geometry, the number of non-bonding domains on the central atom, and the polarity of the molecule. | Homework.Study.com

Draw the Lewis structure for ClF3 and provide the following information. a. formal charge for each atom b. total number of electron domains c. electron geometry d. molecular geometry e. polarity

ClF3 is a polar and planar molecule - Chemistry - Chemical Bonding and Molecular Structure - 13008925 | Meritnation.com